- Last edited on January 25, 2022
Introduction to Benzodiazepines
Primer
Benzodiazepines are a class of psychotropic medications with sedative-hypnotic, anxiolytic, anticonvulsant, and muscle relaxant effects. Benzodiazepines are among the most misused and misprescribed medications in the world.[1][2] Benzodiazepines carry a risk of increased sedation, cognitive impairment, and respiratory depression in those with liver impairments, and there is an increased risk of respiratory depression with opioid or alcohol use.
History
In 1955, chemist Leo Sternbach while working at Hoffmann-La Roche serendipitously identified the first benzodiazepine, chlordiazepoxide (Librium).[3] Initially thought to be less toxic and having little effect on respiratory depression, benzodiazepines became the most commonly prescribed medications in the mid-to-late 1970s. By the 1980s, concerns about addiction and abuse began to come up, particularly for older adults.
Pharmacokinetics
Distribution
- Benzodiazepines are highly protein bound (70-99%) but rapidly distribute to CNS. Benzodiazepines cross the blood brain barrier (BBB) by passive diffusion, and thus, the rate of CNS distribution correlates with lipophilicity.
- Since all benzodiazepines are relatively lipophilic, their degree of lipophilicity influences the duration of clinical effect of the medication.
Metabolism
All benzodiazepines are metabolized by the liver. However, some benzodiazepines (i.e. - lorazepam, oxazepam, and tamazepam) do not go through cytochrome P450 metabolism (Phase I metabolism), and are only metabolized via glucuronidation (Phase II metabolism).
The "LOT" Benzodiazepines
- Lorazepam, oxazepam, and tamazepam are the 3 benzodiazepines that undergo glucuronidation (Phase II metabolism) but not cytochrome p450 metabolism (Phase I metabolism). In patients with advanced liver disease, phase II (glucuronidation) reactions are better preserved. Benzodiazepines undergoing Phase II metabolism also have no active metabolites. Thus, these benzodiazepines can be used in patients with hepatic or renal dysfunction, and the half-life of the medications remain relatively preserved. The mnemonic “LOT” is a way to remember which benzodiazepines are metabolized through glucuronidation.
Cytochrome P450 Metabolized Benzodiazepines
- The rest of the benzodiazepines (e.g. - midazolam, clonazepam, and diazepam) are primarily metabolized via hepatic cytochrome P450 (microsomal oxidation) pathway (CYP3A4). As a result, there are potentially many more drug-drug interactions. These benzodiazepines can have a prolonged duration of effect in patients with severe liver impairment.
Metabolites and Special Populations
- Some benzodiazepines continue to exert their action via their active metabolites.
- For example, diazepam produces the active metabolites oxazepam, desmethyldiazepam, and temazepam.
- These metabolites increase the duration of drug action and can have an impact in older adults and in those with extensive hepatic disease.
- On the other hand, midazolam produces no active metabolites.[4]
Pharmacodynamics
Mechanism of Action
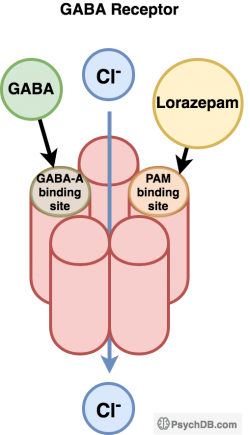
Benzodiazepines (and Z-drugs in general) are positive allosteric modulators (PAMs - this is not the same as agonists!) that act on γ-aminobutyric acid (GABA-A) receptor binding sites that potentiates GABA's inhibitory effect. The combination of GABA at the GABA-A receptor’s agonist site and a benzodiazepine-receptor agonist at the positive allosteric site increases the overall frequency of the chloride channel opening than GABA alone by itself. When a greater frequency of chloride ions to enter through the receptor, this increases polarization and increases GABA-mediated inhibitory action.
Benzodiazepines are highly protein bound, distribute widely in the body, and accumulate in lipid-rich areas such as the central nervous system and fatty tissue. The more lipophilic a benzodiazepine, the higher the rate of absorption and faster the onset of clinical action. For example, diazepam and midazolam have the highest lipid solubility and therefore also the quickest onset of action.
Indications
Most indications for benzodiazepines are based on short-term studies and used in acute medical conditions such as:
- Catatonia (very high doses of benzodiazepines can be used)
- Anticonvulsant action (alcohol withdrawal seizures and seizure disorders)
- Anxiolytic/anti-agitation (rapidly acting and is useful in acute mania or acute psychosis)
- Hypnotic (for short-term use in insomnia)
- Muscle relaxant (e.g. - for antipsychotic-induced acute dystonia)
Comparison of Benzodiazepines (Equivalence Table)
Naming Tip
All benzodiazepines end in -pam or -lam, The only exception are the benzodiazepines chlordiazepoxide and clorazepate.Note that most benzodiazepine equivalence estimates are based on expert clinical opinion, hypothesized equivalence tables (see above links). As a result, these values are meant as a guide and not a direct reflection of equivalence!
Benzodiazepine Equivalence Table
Adapted From: https://www.benzo.org.uk/bzequiv.htm, https://benzoreform.org/comparison-of-benzodiazepine-pharmacology/, and https://clincalc.com/Benzodiazepine/| Benzodiazepines | Diazepam Equivalent (10 mg), PO | Duration of Action | Peak Onset (hours) | Half-life (hours) | Notes |
|---|---|---|---|---|---|
| Alprazolam | 0.5 mg | Short | 0.7-1.6 | 6-20 | High risk of misuse |
| Clobazam | 20 mg | Long | 0.5-4 | 42-47 | Commonly used as adjunctive treatment of seizures associated with Lennox-Gastaut syndrome |
| Clonazepam | ~0.25 mg to 0.5 mg[5] | Long | 1-4 | 18-39 | Fast onset, long acting, high potency, partial serotonin agonist |
| Diazepam | 10 mg | Long | 1 | 20-100 | Commonly used in management of alcohol withdrawal |
| Estazolam | 1-2 mg | Intermediate | 1-6 | 10-24 | May be used in short-term management of insomnia |
| Flurazepam | 15-30 mg | Short | 0.5-2 | 47-100* | May be used in short-term management of insomnia |
| Lorazepam | 1 mg | Intermediate (PO) Short (SL/IV) | 1.-1.5 | 10-20 | Commonly used in pharmacological management of agitation, severe anxiety, alcohol withdrawal, seizure disorders, procedural sedation, insomnia |
| Midazolam | 5 mg[6] | Short | 15 minutes (IM) | 3 | Seizures, procedural sedation, severe agitation |
| Oxazepam | 20 mg | Short | 2-3 | 4-15 | Oxazepam is the active metabolite of diazepam and tamazepam (among others) |
| Tamazepam | 10-20 mg | Short[7] | 0.75-1.5 | 8-22 | - |
| Quazepam | 20 mg | Long | 2 | 25-100 | - |
| Triazolam | 0.5 mg | Short | 0.75-2 | 2 | - |
| Non-Benzodiazepines | |||||
| Zaleplon | 20 mg | Short | 1 | 2 | Used in short-term treatment of insomnia |
| Zolpidem | 20 mg | Short | 1 | 2 | Used in short-term treatment of insomnia |
| Zopiclone | 15 mg | Short | 1-2 | 5-6 | Used in short-term treatment of insomnia |
| Eszopiclone | 3 mg | Short | 1 | 6 | Used in short-term treatment of insomnia |
Mnemonic
TheATOM benzodiazepines all have rapid onset and are short-acting, which increases risk for abuse, misuse, and addiction.A- AlprazolamT- TriazolamO- OxazepamM- Midazolam
Side Effects
- Anterograde amnesia, anticholinergic effects, drowsiness, fatigue, poor concentration, poor coordination, poor psychomotor slowing
Adverse Events
Benzodiazepines carry a higher adverse event rate compared to other psychotropics such as antidepressants.[8][9] Thus, it is particularly important to prescribers to have informed consent discussions with patients about the use of benzodiazepines and their short-term indicatons.
Addiction
- Short-acting, rapid-onset benzodiazepines (e.g. - alprazolam) has much higher addiction potential
- For example, clonazepam and diazepam are considered less addictive than say alprazolam, due to their longer half-lives
- However, clonazepam is also less addictive than diazepam because its onset is delayed, compared to the onset of diazepam, which is rapid.
- Tolerance and withdrawal can develop after minimum 4 weeks of treatment
- Psychological dependence (i.e. - cravings) can develop at any point during treatment
Cognitive Impairment
- Benzodiazepines significantly increase the risk for confusion, delirium, and cognitive impairment in the elderly.
Systemic Effects
Respiration
Sleep
- Benzodiazepines decreases the amount of REM sleep
Deprescribing/Tapering
- A typical benzodiazepine taper is 10 to 20% every 1 to 2 weeks, but may need to be even slower in those who have been on a benzodiazepine for many years.
Withdrawal
Drug-Drug Interactions
Drug-Drug Interactions
| Drug | Interaction |
|---|---|
| Fluoxetine, fluvoxamine, sertraline | Decreases metabolism, and increases plasma levels of benzodiazepines metabolized by CYP3A4 (e.g. - alprazolam, diazepam) |
| Carbamazepine | Increases metabolism and decreases plasma levels |
| Isoniazid | Decreases metabolism, and increases plasma levels of benzodiazepines metabolized by CYP3A4 (e.g. - alprazolam, diazepam) |
| Erythromycin | Decreases metabolism, and increases plasma levels of benzodiazepines metabolized by CYP3A4 (e.g. - alprazolam, diazepam) |